A recent clinical trial indicates that low-dose aspirin may significantly reduce the risk of colorectal cancer recurrence in patients with specific genetic mutations following surgery.
A large-scale clinical trial conducted by researchers at Karolinska Institutet and Karolinska University Hospital in Sweden has revealed promising results regarding the use of low-dose aspirin as a post-operative treatment for patients with colorectal cancer. The study suggests that this common medication may play a critical role in reducing the recurrence of cancer in patients whose tumors exhibit certain genetic alterations.
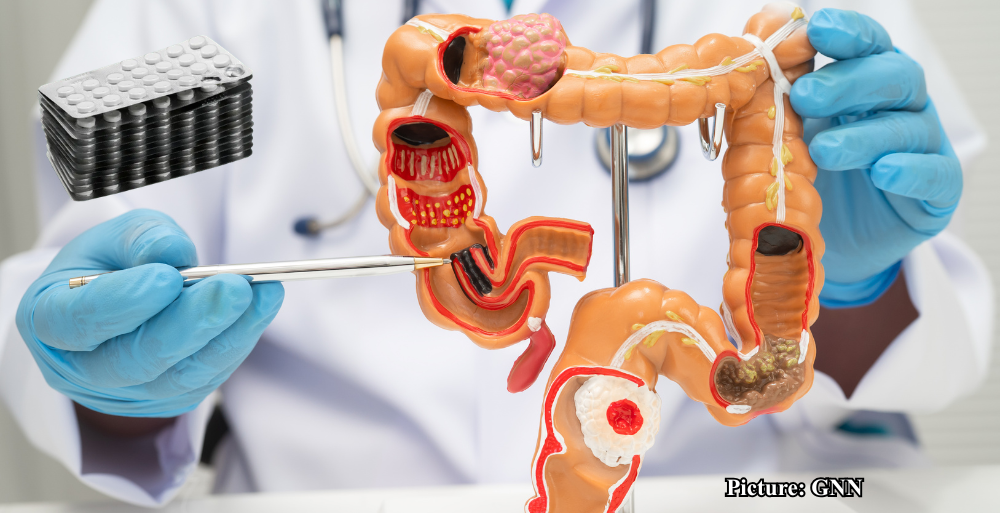
The trial, which involved more than 3,500 participants across 33 hospitals in Sweden, Norway, Denmark, and Finland, focused specifically on patients with colorectal cancer who had undergone surgical treatment. Findings indicate that those who took a daily low dose of aspirin were significantly less likely to experience a return of their cancer, particularly if their tumors had mutations in the PI3K signaling pathway, a critical pathway that influences cell growth and survival.
Trial Results and Recurrence Risk
The randomized, controlled trial assigned participants to receive either 160 mg of aspirin daily or a placebo for a duration of three years post-surgery. Results demonstrated a noteworthy difference in recurrence rates among patients with PIK3CA mutations. Only 7.7 percent of those taking aspirin experienced a recurrence of their cancer, compared to 14.1 percent in the placebo group. Similarly, patients with related genetic alterations saw recurrence rates of 7.7 percent versus 16.8 percent in the placebo group. Overall, the data suggests that aspirin could reduce the risk of cancer recurrence by approximately 50 percent for these patients.
Colorectal cancer remains a significant global health issue, with nearly two million new diagnoses each year. Even after surgical intervention, many patients face a heightened risk of recurrence, particularly if cancer cells have metastasized beyond the original tumor site. According to Anna Martling, the study’s first author and a professor at Karolinska Institutet, “Aspirin is being tested here in a completely new context as a precision medicine treatment. This is a clear example of how we can use genetic information to personalize treatment and at the same time save both resources and suffering.”
Mechanisms Behind Aspirin’s Efficacy
The research team posits that aspirin may exert its beneficial effects through several mechanisms. It has the potential to reduce inflammation, limit platelet activity that can facilitate cancer spread, and interfere with tumor growth. “Although we do not yet fully understand all the molecular links, the findings strongly support the biological rationale and suggest that the treatment may be particularly effective in genetically defined subgroups of patients,” Martling added.
Furthermore, the study indicated that nearly 89 percent of patients treated with aspirin remained cancer-free three years after surgery, in contrast to approximately 79 to 81 percent of those in the placebo group. However, the treatment is not without its drawbacks; severe side effects were reported in 16.8 percent of aspirin users, compared to 11.6 percent among the placebo group, highlighting the necessity for careful consideration of the treatment’s risks.
Implications for Treatment Accessibility
One of the most significant aspects of this study is the accessibility of aspirin as a treatment option. Given that it is widely available and considerably less expensive than many contemporary cancer therapies, the potential incorporation of aspirin into post-operative care represents a promising development in cancer treatment. If future guidelines embrace this approach, genetic testing could serve as a means of identifying patients most likely to benefit from the addition of aspirin to their treatment regimen.
“Aspirin is a drug that is readily available globally and extremely inexpensive compared to many modern cancer drugs, which is very positive,” Martling noted. The implications of this study also extend to healthcare systems and policy makers, as it could lead to more cost-effective treatment strategies in oncology.
Funding and Research Integrity
The study, titled “Low-Dose Aspirin for PI3K-Altered Localized Colorectal Cancer,” was published in the New England Journal of Medicine on September 17, 2025. It was partially funded by the Swedish Research Council and the Swedish Cancer Society. The researchers disclosed no conflicts of interest, reinforcing the integrity of the findings.
The trial’s results not only advance understanding of the role of aspirin in cancer treatment but also open pathways for further investigation into precision medicine, tailoring treatment to the genetic profiles of patients. As research in this area continues to evolve, the potential for integrating common medications like aspirin into standard cancer care could significantly impact patient outcomes and healthcare resources.





No Comment! Be the first one.